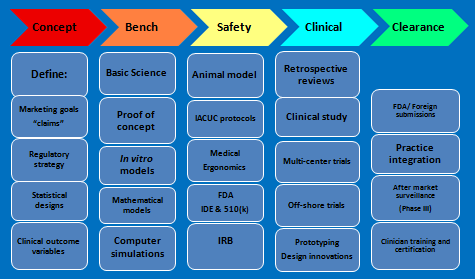
-KEY CAPABILITIES AND SERVICES-
- Basic medical science
- Chart a path to market clearance
- Regulatory affairs
- Clinical studies & trials
- Investigator & subject recruitment
- Validated medical planimetry
- Imaging solutions
- Medical ergonomics
- Design innovation
- Data management
- Bio-statistics
-STEPPING STONES FROM CONCEPT TO CLEARANCE-
-INITIATION-
The first few steps are the most important ones. BMC, Inc., will help you chart the most time- and cost-effective course from concept to the market place based on your unique circumstances.
-RESEARCH-
BMC, Inc. will help you establish the appropriate research projects for your medical device. Anything from literature reviews to university-based controlled clinical trials.
-REGULATORY-
BMC Inc. will prepare and manage submissions to the FDA (devices only, we don't do drugs), Institutional Review Boards (IRB), Animal Care and Use Boards and University Office of Sponsored Projects.
-AFTERMARKET-
BMC, Inc. will design and monitor a program for aftermarket surveillance of safety and efficacy. Training is an essential element in the field of new and innovative medical devices. BMC, Inc. has extensive experience in training clinical personnel and helping your company establish a network of medical gurus for your specific medical nitch.